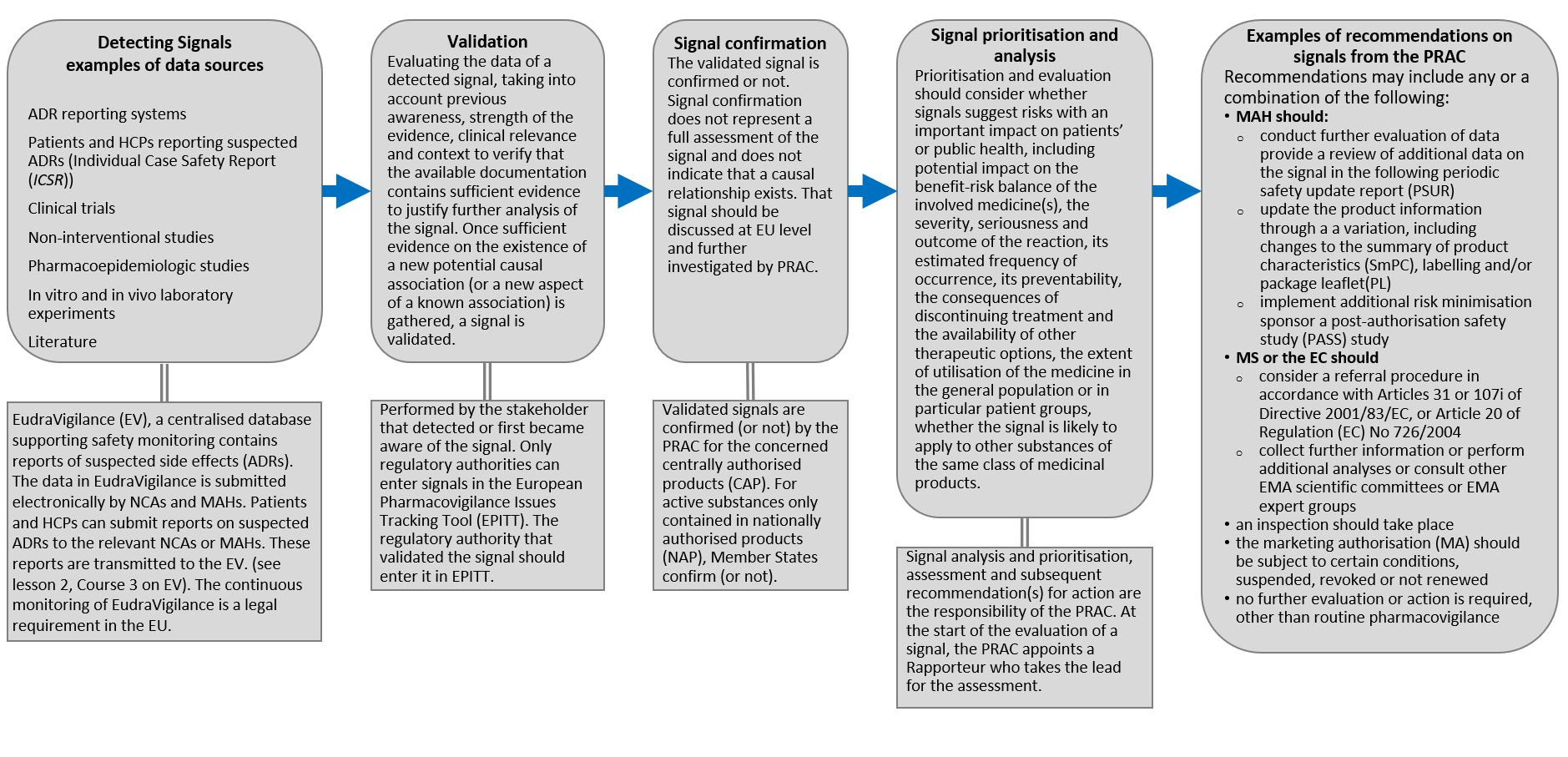
News from the EACPT: New European Medicines Agency advice on black triangle prompts for medicines monitoring

Improving the Safety of Medicines in the European Union: From Signals to Action - Potts - 2020 - Clinical Pharmacology & Therapeutics - Wiley Online Library
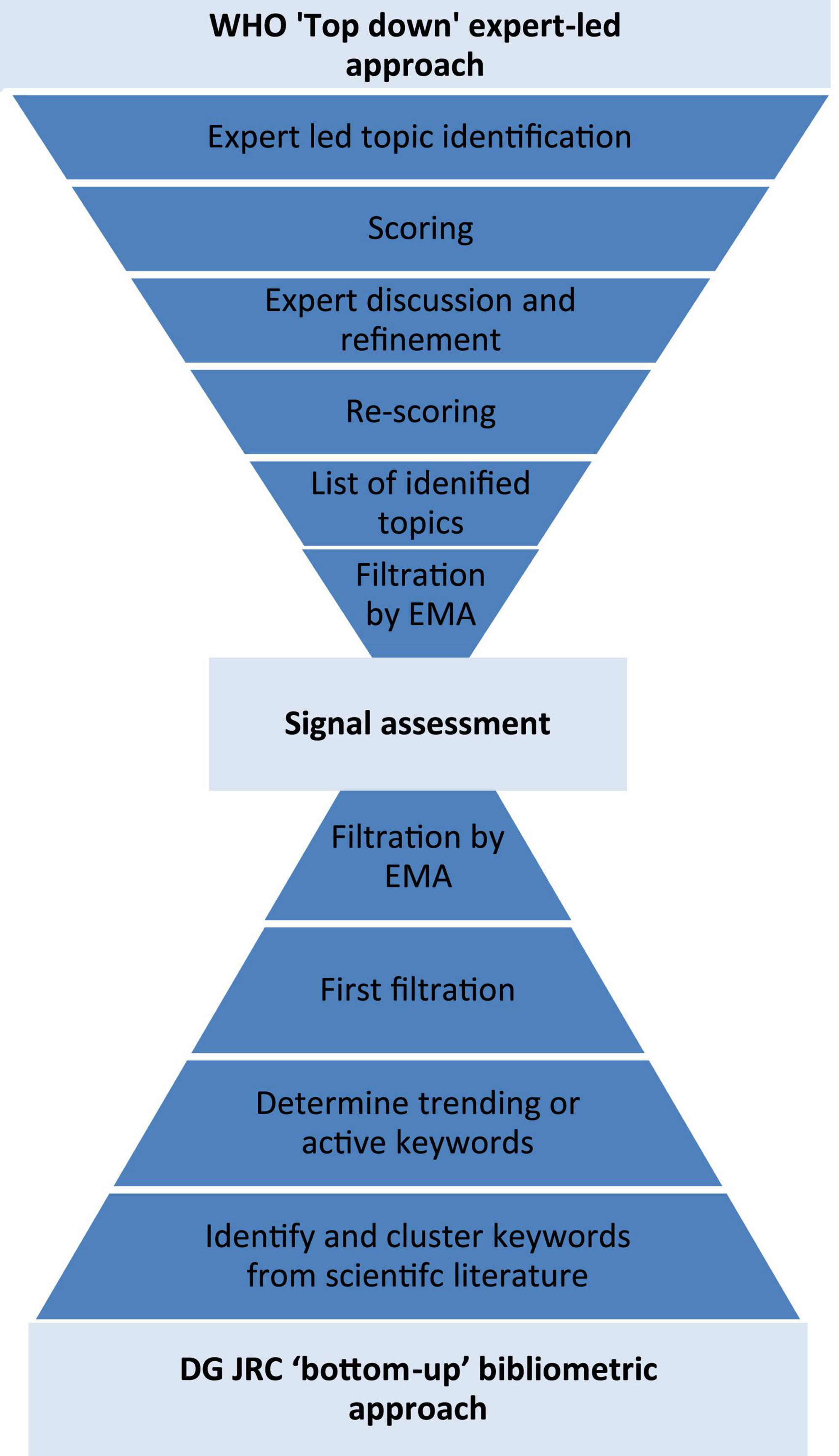
Frontiers | Health horizons: Future trends and technologies from the European Medicines Agency's horizon scanning collaborations

Evaluation of quantitative signal detection in EudraVigilance for orphan drugs: possible risk of false negatives | Semantic Scholar
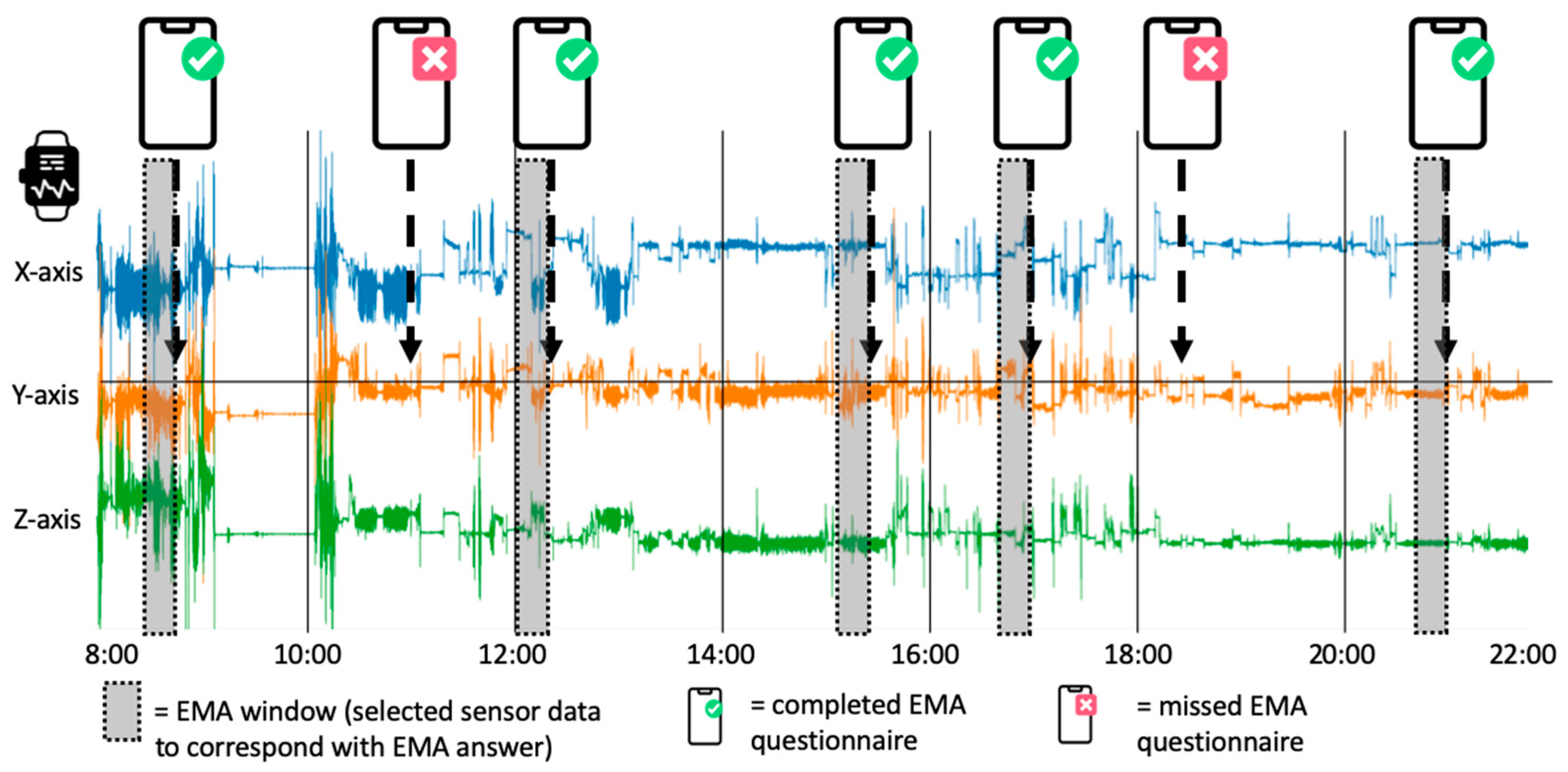
Data | Free Full-Text | A Long-Term, Real-Life Parkinson Monitoring Database Combining Unscripted Objective and Subjective Recordings

Data Safety and Monitoring Boards Should Be Required for Both Early- and Late-Phase Clinical Trials - ScienceDirect