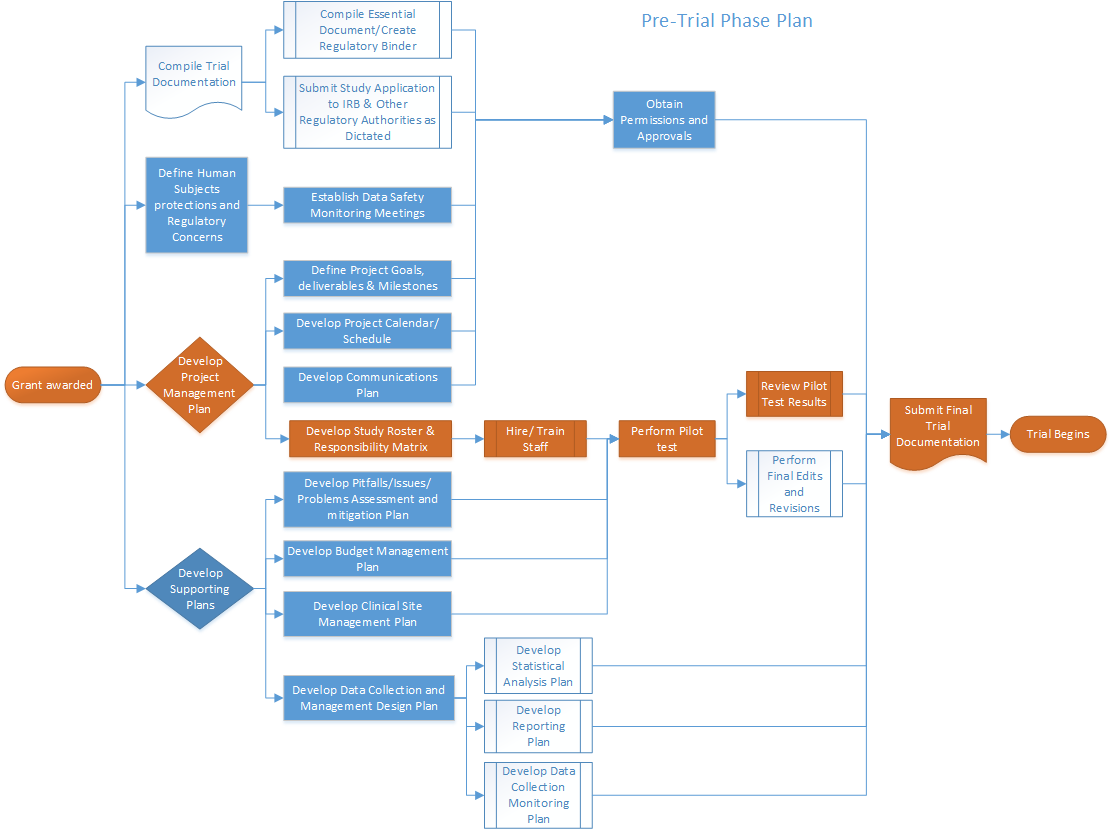
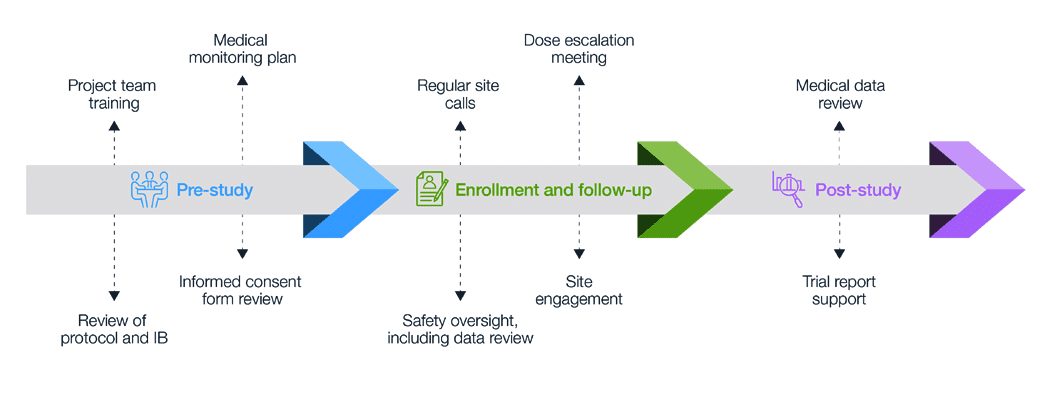
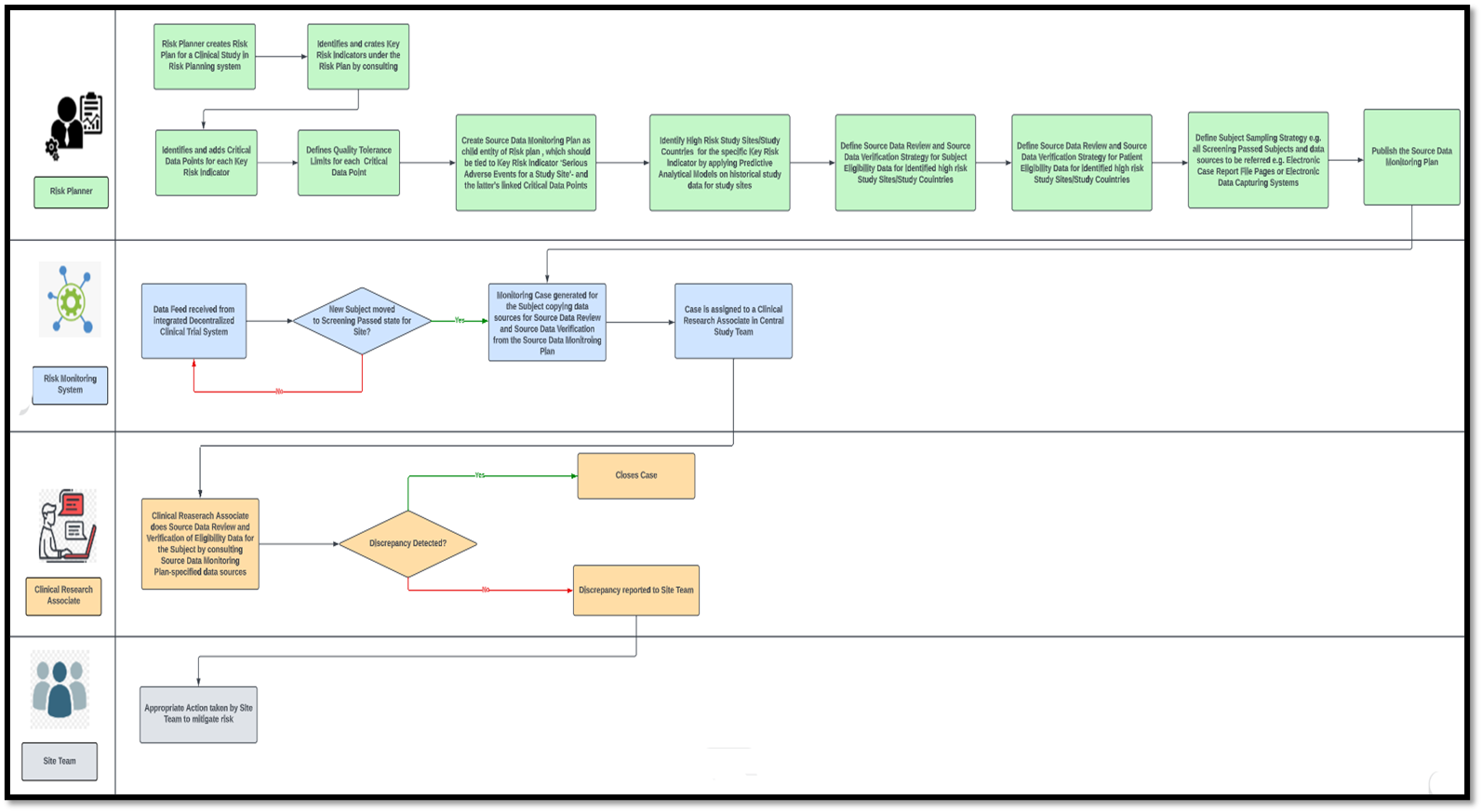
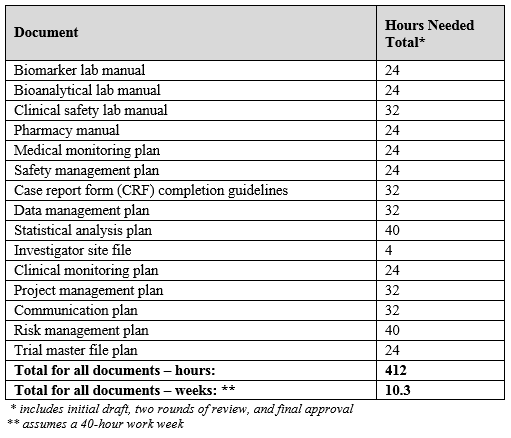
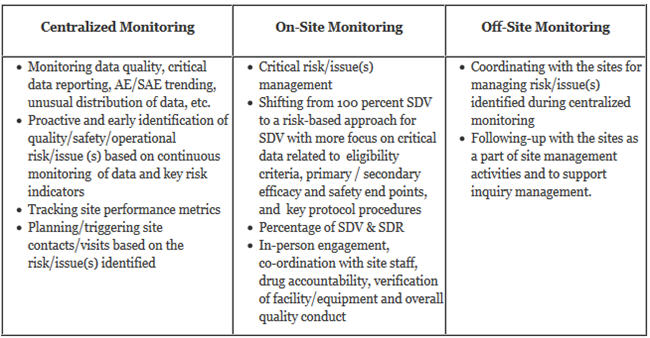
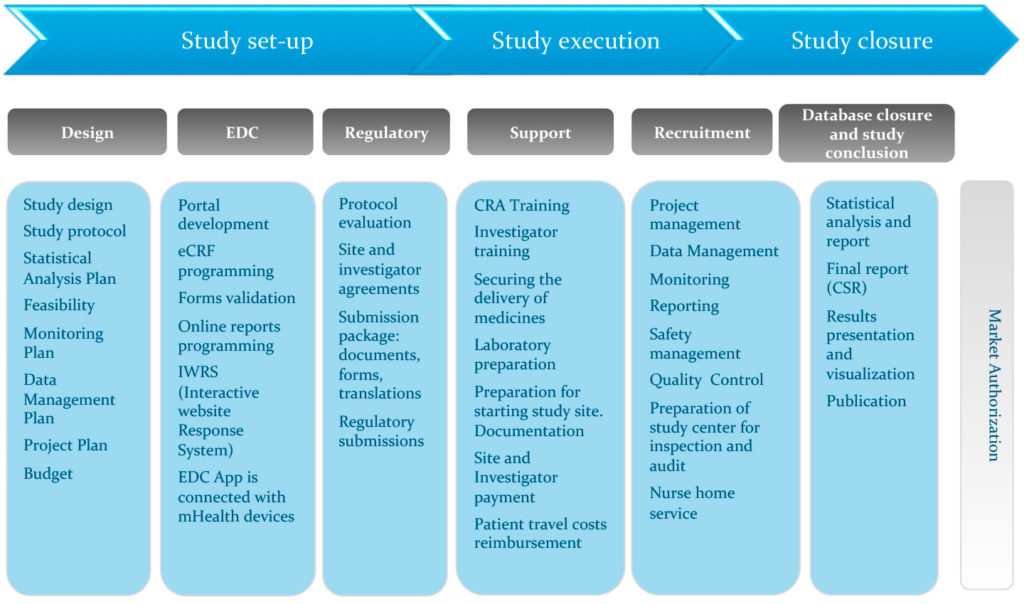
A Centralized Monitoring Approach Using Excel for the Quality Management of Clinical Trials | Applied… | How to plan, Sorority recommendation letter, Support letter

Validation of a risk-assessment scale and a risk-adapted monitoring plan for academic clinical research studies — The Pre-Optimon study - ScienceDirect

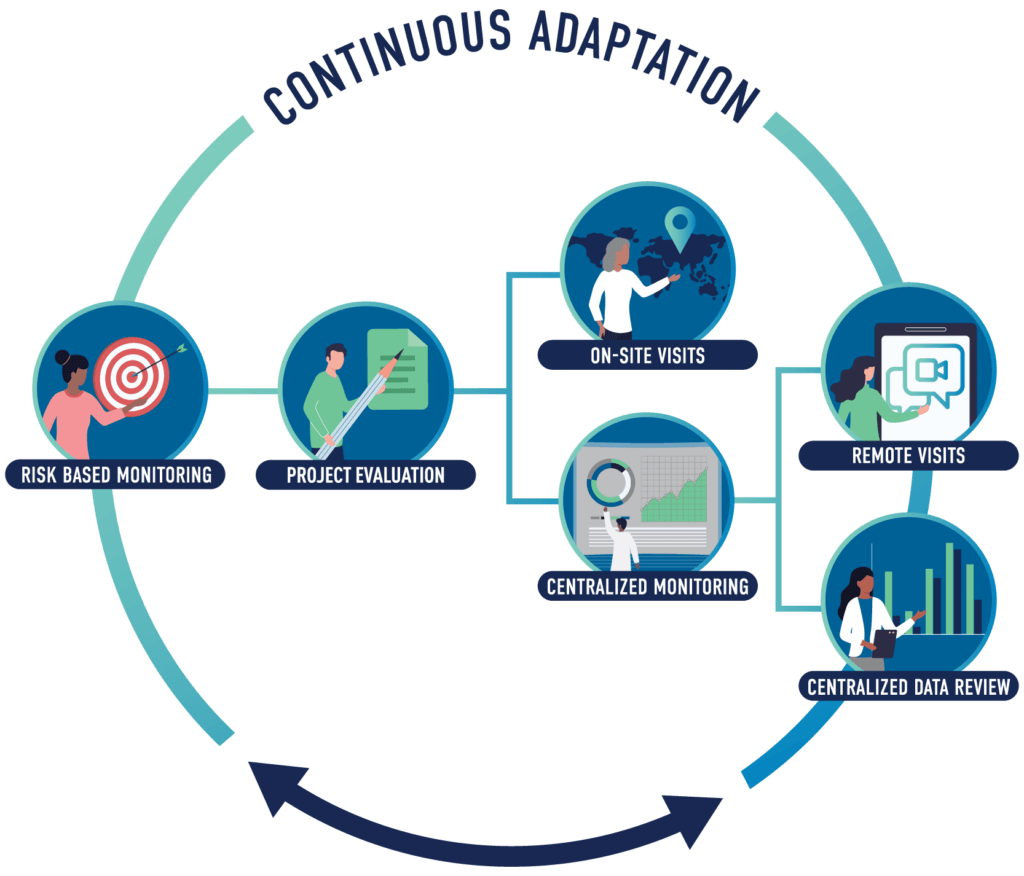
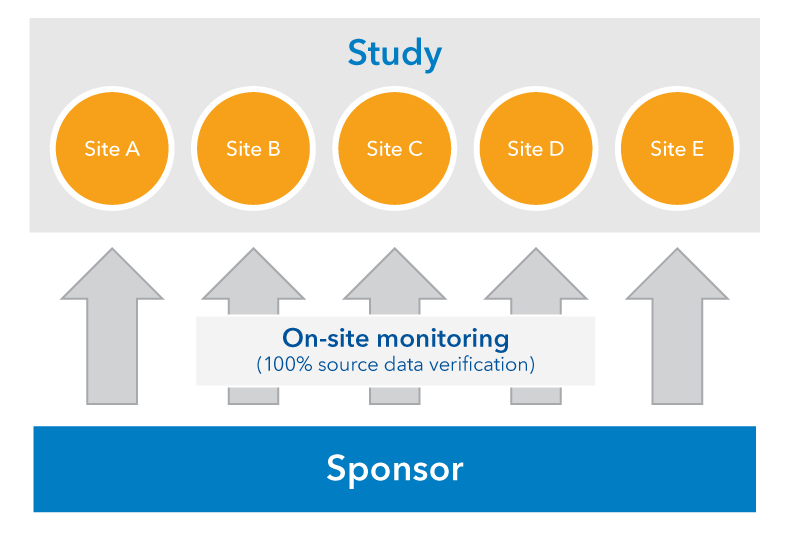
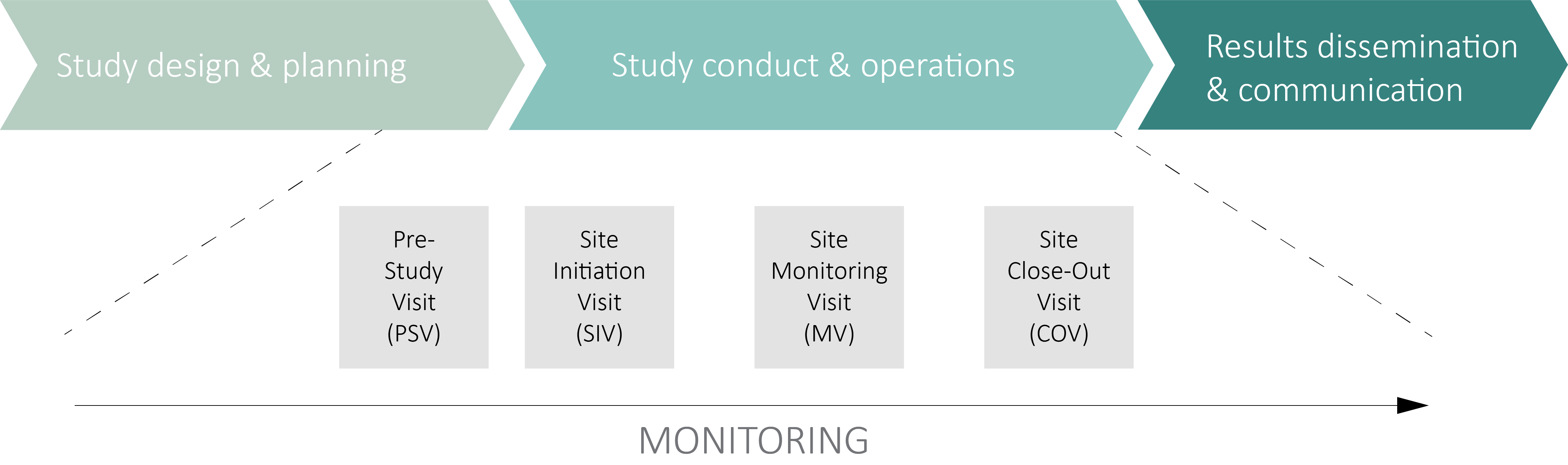
Risk-proportionate clinical trial monitoring: an example approach from a non-commercial trials unit | Trials | Full Text
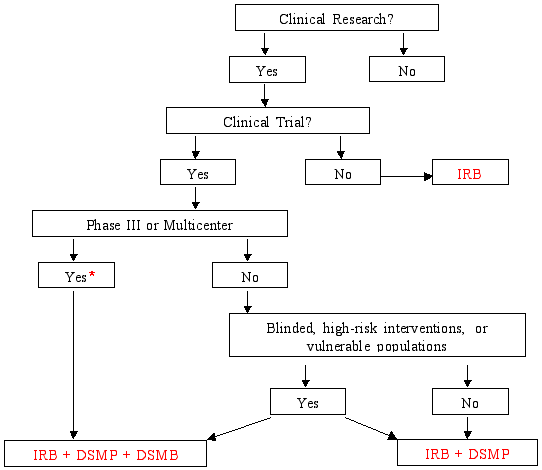
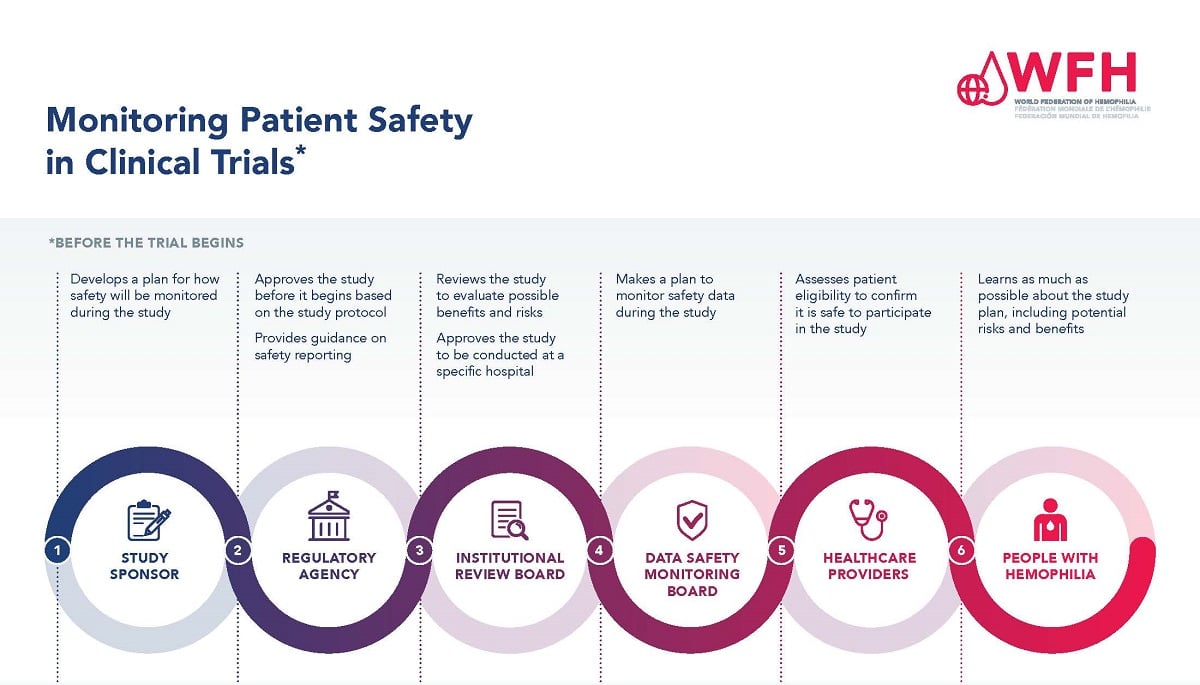
Guidelines for Developing a Data and Safety Monitoring Plan | National Institute on Drug Abuse (NIDA)

Guidelines for Developing a Data and Safety Monitoring Plan | National Institute on Drug Abuse (NIDA)
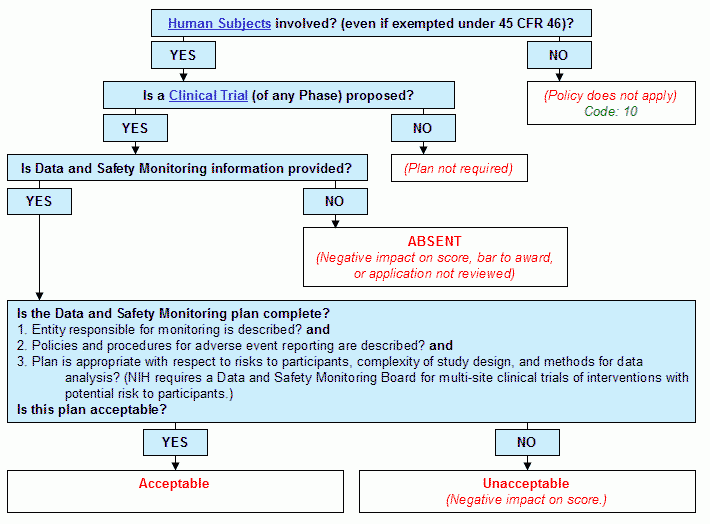
Decision Tree for Data and Safety Monitoring Plan | NIAID: National Institute of Allergy and Infectious Diseases